|
5/31/2023 0 Comments Le chatelier principle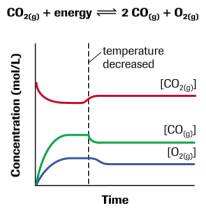
The amount of catalyst does not affect the equilibrium concentration of reactants and products. They require a catalyst to speed them up. There are many chemical reactions that proceed very slowly on their own. Hence, the equilibrium will shift toward the reactant’s side. The system moves in such a way as to decrease the temperature. It increases the temperature of the system. In fact, an increase in temperature favors endothermic reactions, whereas, a decrease in temperature favors exothermic reactions.įor example, the above reaction proceeds with the release of heat energy. In both of the case equilibrium of the system is disturbed. It depends upon whether a chemical reaction is endothermic or exothermic.Īn increase in temperature adds heat to the system whereas a decrease in temperature removes the heat from the system. Effect of change in temperatureĬhemical equilibrium can also be disturbed by changing the temperature. So, a decrease in pressure shift equilibrium toward a forward direction whereas, an increase in pressure shift equilibrium toward a backward direction. It means that the volume of the system is increasing. This reaction proceeds with an increase in the number of moles. It does not affect the equilibria of solid substances or liquids or gases having equal moles of reactants and products.įor example, reactants an d products have an equal number of moles so pressure change does not affect the equilibrium state. By principle, when the pressure is increasing in a gaseous equilibrium reaction, the equilibrium will shift in direction with less number of moles to decrease the pressure. If the reaction proceeds with an increase in the number of moles then the increase in volume shifts the equilibrium backward and vice versa.Ĭhange in pressure only affects the equilibrium state of gaseous substances having unequal moles of product and reactants. Their effect is similar to the effect of pressure change. Effect of change in volumeĬhanges in volume can also influence the equilibrium of a gaseous chemical reaction. This can also be explained by the common ion effect. This will help to maintain dynamic equilibrium. If the concentration of one species is increased at an equilibrium state then the system readjusts in such a way as to decrease the concentration of that species. At that particular time, the system makes adjustments accordingly to minimize the effect of change. When the concentration of the reactant or product is changed, then the equilibrium state of the system is disturbed. The system has an equal concentration of reactant and product at equilibrium. Changing the concentration of reactants or productsĪpplication of Le Chatelier Principle Effect of change in concentrationĪt a dynamic equilibrium state, the rate of the forward reaction is equal to the rate of the reverse reaction.It makes adjustments in such a way as to minimize the effect of disturbance. The Chatelier principle helps to maintain the dynamic equilibrium of the system. For example, change in concentration, temperature, volume, pressure, catalysts, etc. The system can not completely cancel out the effect of these changes but it minimizes the effects of changes. 
There are different parameters that can disturb the dynamic equilibrium state of the system. It was formulated by H.Le Chatelier and F Braun. It is the application of the law of mass action. The Le-chatelier principle is applicable to reversible reactions. This principle can be used to predict how a chemical system will respond to changes in these factors, and it can be used to design experiments to alter the equilibrium state of a system. It is a general rule that describes the effect of changes in temperature, pressure, and concentration on chemical systems that are in equilibrium. According to Le chatelier’s principle, if stress is applied to the system at equilibrium, then the system adjusts itself to overcome applied stress in such a way that the system changes its position, to reduce the effect of that stress.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
